Selected ATcT [1, 2] enthalpy of formation based on version 1.202 of the Thermochemical Network [3]This version of ATcT results[3] was generated by additional expansion of version 1.176 in order to include species related to the thermochemistry of glycine[4].
|
Sulfuric acid hexahydrate | | Formula: (OS(O)(OH)2)(H2O)6 (cr,l) | | CAS RN: 137168-50-4 | | ATcT ID: 137168-50-4*500 | | SMILES: OS(=O)(=O)O.O.O.O.O.O.O | | InChI: InChI=1S/H2O4S.6H2O/c1-5(2,3)4;;;;;;/h(H2,1,2,3,4);6*1H2 | | InChIKey: LSRGDVARLLIAFM-UHFFFAOYSA-N | | Hills Formula: H14O10S1 | 2D Image: | 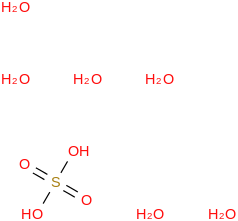 | | Aliases: (OS(O)(OH)2)(H2O)6; Sulfuric acid hexahydrate; Hydrogen sulfate hexahydrate; (O2S(OH)2)(H2O)6; ((O2S)(OH)2)(H2O)6; ((SO2)(OH)2)(H2O)6; (S(O)(O)(OH)2)(H2O)6; (S(O)2(OH)2)(H2O)6; (H2SO4)(H2O)6; H2SO4.6H2O | | Relative Molecular Mass: 206.1712 ± 0.0068 |
| ΔfH°(0 K) | ΔfH°(298.15 K) | Uncertainty | Units |
|---|
| -2588.91 | ± 0.20 | kJ/mol |
| Top contributors to the provenance of ΔfH° of (OS(O)(OH)2)(H2O)6 (cr,l)The 20 contributors listed below account only for 73.2% of the provenance of ΔfH° of (OS(O)(OH)2)(H2O)6 (cr,l).
A total of 150 contributors would be needed to account for 90% of the provenance.
Please note: The list is limited to 20 most important contributors or, if less, a number sufficient to account for 90% of the provenance. The Reference acts as a further link to the relevant references and notes for the measurement. The Measured Quantity is normaly given in the original units; in cases where we have reinterpreted the original measurement, the listed value may differ from that given by the authors. The quoted uncertainty is the a priori uncertainty used as input when constructing the initial Thermochemical Network, and corresponds either to the value proposed by the original authors or to our estimate; if an additional multiplier is given in parentheses immediately after the prior uncertainty, it corresponds to the factor by which the prior uncertainty needed to be multiplied during the ATcT analysis in order to make that particular measurement consistent with the prevailing knowledge contained in the Thermochemical Network.
| Contribution
(%) | TN
ID | Reaction | Measured Quantity | Reference | | 18.3 | 125.2 | 1/2 O2 (g) + H2 (g) → H2O (cr,l) | ΔrH°(298.15 K) = -285.8261 ± 0.040 kJ/mol | Rossini 1939, Rossini 1931, Rossini 1931b, note H2Oa, Rossini 1930 | | 12.9 | 9602.1 | S (cr,l) + O2 (g) → OSO (g) | ΔrH°(298.15 K) = -296.847 ± 0.200 kJ/mol | Eckman 1929, note SO2 | | 8.5 | 9749.1 | S (cr,l) + 3/2 O2 (g) + H2O (cr,l) → OS(O)(OH)2 (aq, 115 H2O) | ΔrH°(298.15 K) = -143.85 ± 0.06 kcal/mol | Good 1960, CODATA Key Vals | | 6.2 | 9749.2 | S (cr,l) + 3/2 O2 (g) + H2O (cr,l) → OS(O)(OH)2 (aq, 115 H2O) | ΔrH°(298.15 K) = -143.92 ± 0.07 kcal/mol | Mansson 1963, CODATA Key Vals | | 5.4 | 2374.7 | CH4 (g) + 2 O2 (g) → CO2 (g) + 2 H2O (cr,l) | ΔrH°(298.15 K) = -890.578 ± 0.078 kJ/mol | Schley 2010 | | 5.0 | 2376.1 | 2 H2 (g) + C (graphite) → CH4 (g) | ΔrG°(1165 K) = 37.521 ± 0.068 kJ/mol | Smith 1946, note COf, 3rd Law | | 2.5 | 9788.1 | S (cr,l) + 3/2 O2 (g) + H2O (cr,l) → OS(O)(OH)2 (aq, 45 H2O) | ΔrH°(298.15 K) = -143.67 ± 0.11 kcal/mol | McCullough 1957a | | 1.7 | 9748.1 | S (cr,l) + 3/2 O2 (g) + H2O (cr,l) → OS(O)(OH)2 (aq, 70 H2O) | ΔrH°(298.15 K) = -143.58 ± 0.09 (×1.477) kcal/mol | McCullough 1953, CODATA Key Vals | | 1.6 | 9587.2 | 2 S (cr,l) → S2 (g) | ΔrG°(570 K) = 9.483 ± 0.138 (×1.874) kcal/mol | Drowart 1968, Detry 1967, 3rd Law | | 1.3 | 115.11 | H2O (g) → O (g) + 2 H (g) | ΔrH°(0 K) = 917.80 ± 0.15 kJ/mol | Thorpe 2021 | | 1.2 | 9587.4 | 2 S (cr,l) → S2 (g) | ΔrG°(600 K) = 8.57 ± 0.29 (×1.044) kcal/mol | Braune 1951, West 1929, Gurvich TPIS, 3rd Law | | 1.2 | 9749.3 | S (cr,l) + 3/2 O2 (g) + H2O (cr,l) → OS(O)(OH)2 (aq, 115 H2O) | ΔrH°(298.15 K) = -143.70 ± 0.07 (×2.278) kcal/mol | Waddington 1956, Mansson 1963, est unc | | 1.1 | 157.1 | OH (g) → [OH]+ (g) | ΔrH°(0 K) = 104989 ± 5 (×2.327) cm-1 | Wiedmann 1992, note unc | | 1.0 | 167.6 | H2O (g) → [OH]+ (g) + H (g) | ΔrH°(0 K) = 18.1183 ± 0.0015 (×1.044) eV | Bodi 2014 | | 0.9 | 152.1 | OH (g) → O (g) + H (g) | ΔrH°(0 K) = 35580 ± 15 cm-1 | Sun 2020 | | 0.9 | 9747.1 | OSO (g) + 1/2 O2 (g) + H2O (cr,l) → OS(O)(OH)2 (cr,l) | ΔrH°(298.15 K) = -231.329 ± 0.040 kJ/mol | NBS Tables 1989 | | 0.8 | 1731.1 | N2 (g) + 3 H2O (cr,l) + 2 H+ (aq) → 3/2 O2 (g) + 2 [NH4]+ (aq) | ΔrH°(298.15 K) = 141.292 ± 0.119 kcal/mol | Vanderzee 1972c | | 0.7 | 169.1 | [OH]- (g) → O- (g) + H (g) | ΔrH°(0 K) = 4.7796 ± 0.0010 (×2.044) eV | Martin 2001, est unc | | 0.7 | 2374.4 | CH4 (g) + 2 O2 (g) → CO2 (g) + 2 H2O (cr,l) | ΔrH°(298.15 K) = -890.61 ± 0.21 kJ/mol | Dale 2002 | | 0.6 | 167.7 | H2O (g) → [OH]+ (g) + H (g) | ΔrH°(0 K) = 18.1190 ± 0.002 eV | Bodi 2014 |
| Top 10 species with enthalpies of formation correlated to the ΔfH° of (OS(O)(OH)2)(H2O)6 (cr,l) | Please note: The correlation coefficients are obtained by renormalizing the off-diagonal elements of the covariance matrix by the corresponding variances.
The correlation coefficient is a number from -1 to 1, with 1 representing perfectly correlated species, -1 representing perfectly anti-correlated species, and 0 representing perfectly uncorrelated species.
| Correlation
Coefficent
(%) | Species Name | Formula | Image | ΔfH°(0 K) | ΔfH°(298.15 K) | Uncertainty | Units | Relative
Molecular
Mass | ATcT ID | | 99.7 | Sulfuric acid heptahydrate | (OS(O)(OH)2)(H2O)7 (cr,l) | 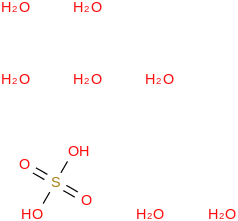 | | -2876.86 | ± 0.22 | kJ/mol | 224.1864 ±
0.0069 | 137168-51-5*500 | | 99.6 | Sulfuric acid pentahydrate | (OS(O)(OH)2)(H2O)5 (cr,l) | 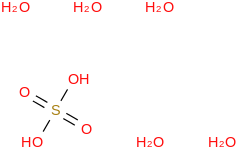 | | -2300.36 | ± 0.18 | kJ/mol | 188.1559 ±
0.0066 | 137168-49-1*500 | | 99.2 | Sulfuric acid octahydrate | (OS(O)(OH)2)(H2O)8 (cr,l) | 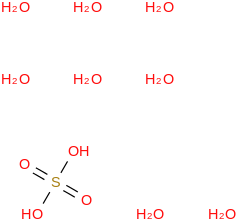 | | -3164.36 | ± 0.24 | kJ/mol | 242.2017 ±
0.0071 | 88434-16-6*500 | | 98.6 | Sulfuric acid nonahydrate | (OS(O)(OH)2)(H2O)9 (cr,l) | 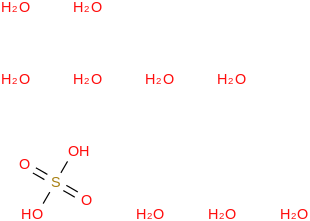 | | -3451.52 | ± 0.25 | kJ/mol | 260.2170 ±
0.0073 | 137168-52-6*500 | | 98.5 | Sulfuric acid tetrahydrate | (OS(O)(OH)2)(H2O)4 (cr,l) | 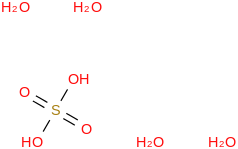 | -2006.93 | -2010.97 | ± 0.17 | kJ/mol | 170.1406 ±
0.0065 | 37006-20-5*500 | | 97.9 | Sulfuric acid decahydrate | (OS(O)(OH)2)(H2O)10 (cr,l) | 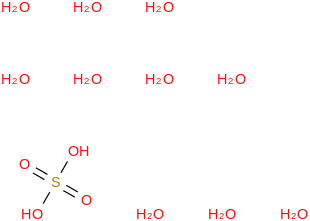 | | -3738.42 | ± 0.27 | kJ/mol | 278.2323 ±
0.0075 | 137168-53-7*500 | | 96.4 | Sulfuric acid dodecahydrate | (OS(O)(OH)2)(H2O)12 (cr,l) | 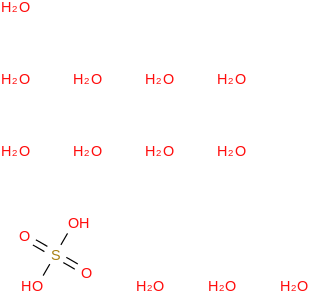 | | -4311.63 | ± 0.31 | kJ/mol | 314.2628 ±
0.0079 | 30383-99-4*500 | | 96.2 | Sulfuric acid trihydrate | (OS(O)(OH)2)(H2O)3 (cr,l) | 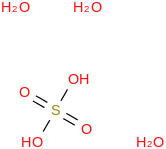 | -1716.08 | -1720.20 | ± 0.15 | kJ/mol | 152.1253 ±
0.0064 | 40835-65-2*500 | | 94.4 | Sulfuric acid pentadecahydrate | (OS(O)(OH)2)(H2O)15 (cr,l) | 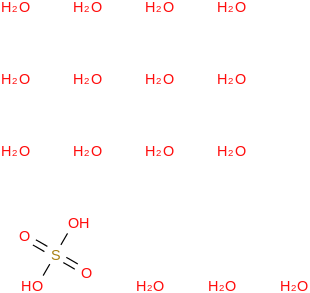 | | -5170.52 | ± 0.37 | kJ/mol | 368.3087 ±
0.0086 | 425605-43-2*500 | | 91.9 | Sulfuric acid dihydrate | (OS(O)(OH)2)(H2O)2 (cr,l) | 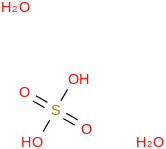 | -1422.14 | -1426.93 | ± 0.14 | kJ/mol | 134.1100 ±
0.0063 | 13451-10-0*500 |
| Most Influential reactions involving (OS(O)(OH)2)(H2O)6 (cr,l)Please note: The list, which is based on a hat (projection) matrix analysis, is limited to no more than 20 largest influences.
|
|
|
|
References
|
|
1
|
|
B. Ruscic, R. E. Pinzon, M. L. Morton, G. von Laszewski, S. Bittner, S. G. Nijsure, K. A. Amin, M. Minkoff, and A. F. Wagner,
Introduction to Active Thermochemical Tables: Several "Key" Enthalpies of Formation Revisited.
J. Phys. Chem. A 108, 9979-9997 (2004)
[DOI: 10.1021/jp047912y]
|
|
2
|
|
B. Ruscic, R. E. Pinzon, G. von Laszewski, D. Kodeboyina, A. Burcat, D. Leahy, D. Montoya, and A. F. Wagner,
Active Thermochemical Tables: Thermochemistry for the 21st Century.
J. Phys. Conf. Ser. 16, 561-570 (2005)
[DOI: 10.1088/1742-6596/16/1/078]
|
|
3
|
|
B. Ruscic and D. H. Bross,
Active Thermochemical Tables (ATcT) values based on ver. 1.202 of the Thermochemical Network (2024); available at ATcT.anl.gov |
|
4
|
|
B. Ruscic and D. H. Bross
Accurate and Reliable Thermochemistry by Data Analysis of Complex Thermochemical Networks using Active Thermochemical Tables: The Case of Glycine Thermochemistry
Faraday Discuss. (in press) (2024)
[DOI: 10.1039/D4FD00110A]
|
|
5
|
|
B. Ruscic,
Uncertainty Quantification in Thermochemistry, Benchmarking Electronic Structure Computations, and Active Thermochemical Tables.
Int. J. Quantum Chem. 114, 1097-1101 (2014)
[DOI: 10.1002/qua.24605]
|
|
6
|
|
B. Ruscic and D. H. Bross,
Thermochemistry
Computer Aided Chem. Eng. 45, 3-114 (2019)
[DOI: 10.1016/B978-0-444-64087-1.00001-2]
|
|
|
|
Formula
|
|
The aggregate state is given in parentheses following the formula, such as: g - gas-phase, cr - crystal, l - liquid, etc.
|
|
|
Uncertainties
|
The listed uncertainties correspond to estimated 95% confidence limits, as customary in thermochemistry (see, for example, Ruscic [5] and Ruscic and Bross[6]).
Note that an uncertainty of ± 0.000 kJ/mol indicates that the estimated uncertainty is < ± 0.0005 kJ/mol.
|
|
|
Website Functionality Credits
|
The reorganization of the website was developed and implemented by David H. Bross (ANL).
The find function is based on the complete Species Dictionary entries for the appropriate version of the ATcT TN.
The molecule images are rendered by Indigo-depict.
The XYZ renderings are based on Jmol: an open-source Java viewer for chemical structures in 3D. http://www.jmol.org/.
|
|
|
Acknowledgement
|
|
This work was supported by the U.S. Department of Energy, Office of Science, Office of Basic Energy Sciences, Division of Chemical Sciences, Geosciences and Biosciences under Contract No. DE-AC02-06CH11357.
|
|
Page generated: 04/09/2025
|







