Selected ATcT [1, 2] enthalpy of formation based on version 1.176 of the Thermochemical Network [3]This version of ATcT results[3] was generated by additional expansion of version 1.172 to include species related to Criegee intermediates that are involved in several ongoing studies[4].
|
Anthracene |
| Formula: C6H4(CH(CC(C4H4)CH)) (cr,l) |
| CAS RN: 120-12-7 |
| ATcT ID: 120-12-7*500 |
| SMILES: c1ccc2cc3ccccc3cc2c1 |
| InChI: InChI=1S/C14H10/c1-2-6-12-10-14-8-4-3-7-13(14)9-11(12)5-1/h1-10H |
| InChIKey: MWPLVEDNUUSJAV-UHFFFAOYSA-N |
| Hills Formula: C14H10 |
2D Image: |
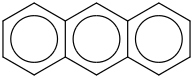 |
| Aliases: C6H4(CH(CC(C4H4)CH)); Anthracene; Anthracin; Green Oil; NSC 7958; Paranaphthalene; Tetra Olive N2G |
| Relative Molecular Mass: 178.2292 ± 0.0112 |
| ΔfH°(0 K) | ΔfH°(298.15 K) | Uncertainty | Units |
|---|
| 150.62 | 124.81 | ± 0.88 | kJ/mol |
|
Top contributors to the provenance of ΔfH° of C6H4(CH(CC(C4H4)CH)) (cr,l)The 20 contributors listed below account only for 73.8% of the provenance of ΔfH° of C6H4(CH(CC(C4H4)CH)) (cr,l).
A total of 41 contributors would be needed to account for 90% of the provenance.
Please note: The list is limited to 20 most important contributors or, if less, a number sufficient to account for 90% of the provenance. The Reference acts as a further link to the relevant references and notes for the measurement. The Measured Quantity is normaly given in the original units; in cases where we have reinterpreted the original measurement, the listed value may differ from that given by the authors. The quoted uncertainty is the a priori uncertainty used as input when constructing the initial Thermochemical Network, and corresponds either to the value proposed by the original authors or to our estimate; if an additional multiplier is given in parentheses immediately after the prior uncertainty, it corresponds to the factor by which the prior uncertainty needed to be multiplied during the ATcT analysis in order to make that particular measurement consistent with the prevailing knowledge contained in the Thermochemical Network.
|
Contribution
(%) | TN
ID | Reaction | Measured Quantity | Reference | | 15.2 | 9227.1 | C6H4(CH(CC(C4H4)CH)) (cr,l) + 33/2 O2 (g) → 14 CO2 (g) + 5 H2O (cr,l) | ΔrH°(298.15 K) = -7062.6 ± 2.1 kJ/mol | Ribeiro da Silva 2007 | | 13.9 | 9236.1 | C6H4(CH(CC(C4H4)CH)) (cr,l) → C6H4(C2H2(CC(C4H4))) (cr,l) | ΔrH°(298.15 K) = -3.11 ± 0.4 kcal/mol | Coleman 1966, est unc | | 10.7 | 9227.2 | C6H4(CH(CC(C4H4)CH)) (cr,l) + 33/2 O2 (g) → 14 CO2 (g) + 5 H2O (cr,l) | ΔrH°(298.15 K) = -7065.0 ± 1.1 (×2.278) kJ/mol | Nagano 2001 | | 4.7 | 9234.1 | C6H4(C2H2(CC(C4H4))) (cr,l) + 33/2 O2 (g) → 14 CO2 (g) + 5 H2O (cr,l) | ΔrH°(298.15 K) = -7048.5 ± 0.9 kJ/mol | Nagano 2002 | | 3.8 | 9227.13 | C6H4(CH(CC(C4H4)CH)) (cr,l) + 33/2 O2 (g) → 14 CO2 (g) + 5 H2O (cr,l) | ΔrH°(298.15 K) = -1687.10 ± 1.0 kcal/mol | Beckers 1931, est unc | | 2.7 | 9227.4 | C6H4(CH(CC(C4H4)CH)) (cr,l) + 33/2 O2 (g) → 14 CO2 (g) + 5 H2O (cr,l) | ΔrH°(298.15 K) = -1689.15 ± 0.41 (×2.89) kcal/mol | Coleman 1966 | | 2.4 | 9227.3 | C6H4(CH(CC(C4H4)CH)) (cr,l) + 33/2 O2 (g) → 14 CO2 (g) + 5 H2O (cr,l) | ΔrH°(298.15 K) = -7063.65 ± 5.28 kJ/mol | Metzger 1983 | | 2.2 | 9236.2 | C6H4(CH(CC(C4H4)CH)) (cr,l) → C6H4(C2H2(CC(C4H4))) (cr,l) | ΔrH°(298.15 K) = -3.0 ± 1.0 kcal/mol | Bender 1952, est unc | | 1.9 | 9225.6 | C6H4(CH(CC(C4H4)CH)) (g) → 14 C (g) + 10 H (g) | ΔrH°(0 K) = 11866.6 ± 5.2 kJ/mol | Karton 2021, Karton 2012a | | 1.7 | 9234.2 | C6H4(C2H2(CC(C4H4))) (cr,l) + 33/2 O2 (g) → 14 CO2 (g) + 5 H2O (cr,l) | ΔrH°(298.15 K) = -7048.1 ± 1.5 kJ/mol | Steele 1990 | | 1.7 | 9227.6 | C6H4(CH(CC(C4H4)CH)) (cr,l) + 33/2 O2 (g) → 14 CO2 (g) + 5 H2O (cr,l) | ΔrH°(298.15 K) = -1688.3 ± 1.5 kcal/mol | Bender 1952, est unc | | 1.5 | 2228.7 | C (graphite) + O2 (g) → CO2 (g) | ΔrH°(298.15 K) = -393.464 ± 0.024 kJ/mol | Hawtin 1966, note CO2e | | 1.5 | 9230.2 | C6H4(C2H2(CC(C4H4))) (g) → C6H4(CH(CC(C4H4)CH)) (g) | ΔrH°(0 K) = 5.65 ± 1.0 kcal/mol | Ruscic G4 | | 1.5 | 9230.4 | C6H4(C2H2(CC(C4H4))) (g) → C6H4(CH(CC(C4H4)CH)) (g) | ΔrH°(0 K) = 5.70 ± 1.0 kcal/mol | Ruscic CBS-n | | 1.3 | 9226.4 | C6H4(CH(CC(C4H4)CH)) (g) + C6H6 (g) → 2 C6H4(C4H4) (g) | ΔrH°(0 K) = -2.87 ± 0.90 kcal/mol | Ruscic CBS-n | | 1.3 | 9226.2 | C6H4(CH(CC(C4H4)CH)) (g) + C6H6 (g) → 2 C6H4(C4H4) (g) | ΔrH°(0 K) = -3.55 ± 0.90 kcal/mol | Ruscic G4 | | 1.3 | 9226.1 | C6H4(CH(CC(C4H4)CH)) (g) + C6H6 (g) → 2 C6H4(C4H4) (g) | ΔrH°(0 K) = -3.63 ± 0.90 kcal/mol | Ruscic G3X | | 1.3 | 9227.12 | C6H4(CH(CC(C4H4)CH)) (cr,l) + 33/2 O2 (g) → 14 CO2 (g) + 5 H2O (cr,l) | ΔrH°(298.15 K) = -1686.3 ± 1.0 (×1.719) kcal/mol | Burriel 1931, as quoted by NIST WebBook, est unc | | 1.2 | 9230.7 | C6H4(C2H2(CC(C4H4))) (g) → C6H4(CH(CC(C4H4)CH)) (g) | ΔrH°(0 K) = 24.0 ± 4.6 kJ/mol | Karton 2021 | | 1.2 | 9230.1 | C6H4(C2H2(CC(C4H4))) (g) → C6H4(CH(CC(C4H4)CH)) (g) | ΔrH°(0 K) = 5.68 ± 1.1 kcal/mol | Ruscic G3X |
|
Top 10 species with enthalpies of formation correlated to the ΔfH° of C6H4(CH(CC(C4H4)CH)) (cr,l) |
Please note: The correlation coefficients are obtained by renormalizing the off-diagonal elements of the covariance matrix by the corresponding variances.
The correlation coefficient is a number from -1 to 1, with 1 representing perfectly correlated species, -1 representing perfectly anti-correlated species, and 0 representing perfectly uncorrelated species.
|
Correlation
Coefficent
(%) | Species Name | Formula | Image | ΔfH°(0 K) | ΔfH°(298.15 K) | Uncertainty | Units | Relative
Molecular
Mass | ATcT ID | | 86.7 | Anthracene | C6H4(CH(CC(C4H4)CH)) (g) | 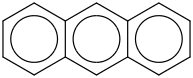 | 255.99 | 226.97 | ± 0.92 | kJ/mol | 178.2292 ±
0.0112 | 120-12-7*0 | | 39.2 | Phenanthrene | C6H4(C2H2(CC(C4H4))) (cr,l) | 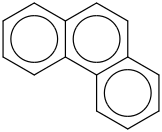 | 136.27 | 110.99 | ± 0.64 | kJ/mol | 178.2292 ±
0.0112 | 85-01-8*500 | | 38.0 | Phenanthrene | C6H4(C2H2(CC(C4H4))) (g) | 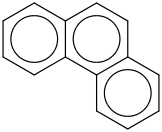 | 231.97 | 203.16 | ± 0.75 | kJ/mol | 178.2292 ±
0.0112 | 85-01-8*0 | | 35.5 | Naphthalene | C6H4(C4H4) (g) | 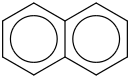 | 171.16 | 147.69 | ± 0.55 | kJ/mol | 128.1705 ±
0.0080 | 91-20-3*0 | | 35.2 | Naphtalene cation | [C6H4(C4H4)]+ (g) | ![c1c(cc2ccccc2c1)[H+]](../images/2992.png) | 956.92 | 933.85 | ± 0.55 | kJ/mol | 128.1700 ±
0.0080 | 34512-27-1*0 | | 34.8 | Naphthalene | C6H4(C4H4) (cr,l) | 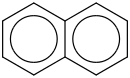 | 94.71 | 75.12 | ± 0.55 | kJ/mol | 128.1705 ±
0.0080 | 91-20-3*500 | | 21.4 | Carbon dioxide | CO2 (g) | 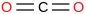 | -393.111 | -393.478 | ± 0.015 | kJ/mol | 44.00950 ±
0.00100 | 124-38-9*0 | | 21.1 | Carbon dioxide cation | [CO2]+ (g) | =O](../images/488.png) | 936.089 | 936.924 | ± 0.017 | kJ/mol | 44.00895 ±
0.00100 | 12181-61-2*0 | | 20.2 | Carbonic acid | C(O)(OH)2 (aq, undissoc) | 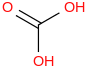 | | -698.673 | ± 0.028 | kJ/mol | 62.0248 ±
0.0012 | 463-79-6*1000 | | 17.8 | Benzoic acid | C6H5C(O)OH (cr,l) | 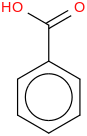 | -367.34 | -384.75 | ± 0.17 | kJ/mol | 122.1213 ±
0.0056 | 65-85-0*500 |
|
Most Influential reactions involving C6H4(CH(CC(C4H4)CH)) (cr,l)Please note: The list, which is based on a hat (projection) matrix analysis, is limited to no more than 20 largest influences.
|
Influence
Coefficient | TN
ID | Reaction | Measured Quantity | Reference | | 0.357 | 9228.10 | C6H4(CH(CC(C4H4)CH)) (cr,l) → C6H4(CH(CC(C4H4)CH)) (g) | ΔrH°(392.5 K) = 23.826 ± 0.186 kcal/mol | Malaspina 1973, 2nd Law | | 0.262 | 9236.1 | C6H4(CH(CC(C4H4)CH)) (cr,l) → C6H4(C2H2(CC(C4H4))) (cr,l) | ΔrH°(298.15 K) = -3.11 ± 0.4 kcal/mol | Coleman 1966, est unc | | 0.163 | 9227.1 | C6H4(CH(CC(C4H4)CH)) (cr,l) + 33/2 O2 (g) → 14 CO2 (g) + 5 H2O (cr,l) | ΔrH°(298.15 K) = -7062.6 ± 2.1 kJ/mol | Ribeiro da Silva 2007 | | 0.114 | 9227.2 | C6H4(CH(CC(C4H4)CH)) (cr,l) + 33/2 O2 (g) → 14 CO2 (g) + 5 H2O (cr,l) | ΔrH°(298.15 K) = -7065.0 ± 1.1 (×2.278) kJ/mol | Nagano 2001 | | 0.077 | 9228.4 | C6H4(CH(CC(C4H4)CH)) (cr,l) → C6H4(CH(CC(C4H4)CH)) (g) | ΔrH°(350.4 K) = 99.0 ± 0.8 (×2.089) kJ/mol | Ribeiro da Silva 2006, note unc | | 0.074 | 9228.5 | C6H4(CH(CC(C4H4)CH)) (cr,l) → C6H4(CH(CC(C4H4)CH)) (g) | ΔrH°(298.15 K) = 100.8 ± 1.7 kJ/mol | Santos 2004 | | 0.063 | 9228.11 | C6H4(CH(CC(C4H4)CH)) (cr,l) → C6H4(CH(CC(C4H4)CH)) (g) | ΔrH°(392.5 K) = 24.148 ± 0.110 (×4) kcal/mol | Malaspina 1973, 2nd Law | | 0.054 | 9228.9 | C6H4(CH(CC(C4H4)CH)) (cr,l) → C6H4(CH(CC(C4H4)CH)) (g) | ΔrH°(349 K) = 100.6 ± 2.0 kJ/mol | de Kruif 1980, note unc | | 0.049 | 9228.14 | C6H4(CH(CC(C4H4)CH)) (cr,l) → C6H4(CH(CC(C4H4)CH)) (g) | ΔrH°(388 K) = 24.1 ± 0.5 kcal/mol | Klochov 1958, Malaspina 1973, Roux 2008, est unc | | 0.049 | 9228.15 | C6H4(CH(CC(C4H4)CH)) (cr,l) → C6H4(CH(CC(C4H4)CH)) (g) | ΔrH°(346.2 K) = 24.3 ± 0.5 kcal/mol | Bradley 1953b, est unc | | 0.045 | 9228.12 | C6H4(CH(CC(C4H4)CH)) (cr,l) → C6H4(CH(CC(C4H4)CH)) (g) | ΔrH°(350.7 K) = 23.54 ± 0.5 (×1.044) kcal/mol | Kelley 1964, est unc | | 0.042 | 9236.2 | C6H4(CH(CC(C4H4)CH)) (cr,l) → C6H4(C2H2(CC(C4H4))) (cr,l) | ΔrH°(298.15 K) = -3.0 ± 1.0 kcal/mol | Bender 1952, est unc | | 0.041 | 9227.13 | C6H4(CH(CC(C4H4)CH)) (cr,l) + 33/2 O2 (g) → 14 CO2 (g) + 5 H2O (cr,l) | ΔrH°(298.15 K) = -1687.10 ± 1.0 kcal/mol | Beckers 1931, est unc | | 0.039 | 9228.13 | C6H4(CH(CC(C4H4)CH)) (cr,l) → C6H4(CH(CC(C4H4)CH)) (g) | ΔrH°(338 K) = 24.7 ± 0.5 (×1.114) kcal/mol | Hoyer 1958, Roux 2008 | | 0.032 | 9228.8 | C6H4(CH(CC(C4H4)CH)) (cr,l) → C6H4(CH(CC(C4H4)CH)) (g) | ΔrH°(338 K) = 102.6 ± 2.6 kJ/mol | Hansen 1986, Roux 2008 | | 0.029 | 9227.4 | C6H4(CH(CC(C4H4)CH)) (cr,l) + 33/2 O2 (g) → 14 CO2 (g) + 5 H2O (cr,l) | ΔrH°(298.15 K) = -1689.15 ± 0.41 (×2.89) kcal/mol | Coleman 1966 | | 0.027 | 9228.6 | C6H4(CH(CC(C4H4)CH)) (cr,l) → C6H4(CH(CC(C4H4)CH)) (g) | ΔrH°(340.5 K) = 100.0 ± 2.8 kJ/mol | Oja 1998 | | 0.025 | 9227.3 | C6H4(CH(CC(C4H4)CH)) (cr,l) + 33/2 O2 (g) → 14 CO2 (g) + 5 H2O (cr,l) | ΔrH°(298.15 K) = -7063.65 ± 5.28 kJ/mol | Metzger 1983 | | 0.022 | 9236.5 | C6H4(CH(CC(C4H4)CH)) (cr,l) → C6H4(C2H2(CC(C4H4))) (cr,l) | ΔrH°(298.15 K) = -4.65 ± 0.6 (×2.278) kcal/mol | Fries 1935, as quoted by Cox 1970, est unc | | 0.019 | 9228.3 | C6H4(CH(CC(C4H4)CH)) (cr,l) → C6H4(CH(CC(C4H4)CH)) (g) | ΔrH°(335 K) = 98.51 ± 3.32 kJ/mol | Goldfarb 2008 |
|
|
|
References
|
|
1
|
|
B. Ruscic, R. E. Pinzon, M. L. Morton, G. von Laszewski, S. Bittner, S. G. Nijsure, K. A. Amin, M. Minkoff, and A. F. Wagner,
Introduction to Active Thermochemical Tables: Several "Key" Enthalpies of Formation Revisited.
J. Phys. Chem. A 108, 9979-9997 (2004)
[DOI: 10.1021/jp047912y]
|
|
2
|
|
B. Ruscic, R. E. Pinzon, G. von Laszewski, D. Kodeboyina, A. Burcat, D. Leahy, D. Montoya, and A. F. Wagner,
Active Thermochemical Tables: Thermochemistry for the 21st Century.
J. Phys. Conf. Ser. 16, 561-570 (2005)
[DOI: 10.1088/1742-6596/16/1/078]
|
|
3
|
|
B. Ruscic and D. H. Bross,
Active Thermochemical Tables (ATcT) values based on ver. 1.176 of the Thermochemical Network (2024); available at ATcT.anl.gov |
|
4
|
|
T. L. Nguyen et al, ongoing studies (2024)
|
|
5
|
|
B. Ruscic,
Uncertainty Quantification in Thermochemistry, Benchmarking Electronic Structure Computations, and Active Thermochemical Tables.
Int. J. Quantum Chem. 114, 1097-1101 (2014)
[DOI: 10.1002/qua.24605]
|
|
6
|
|
B. Ruscic and D. H. Bross,
Thermochemistry
Computer Aided Chem. Eng. 45, 3-114 (2019)
[DOI: 10.1016/B978-0-444-64087-1.00001-2]
|
|
|
|
Formula
|
|
The aggregate state is given in parentheses following the formula, such as: g - gas-phase, cr - crystal, l - liquid, etc.
|
|
|
Uncertainties
|
The listed uncertainties correspond to estimated 95% confidence limits, as customary in thermochemistry (see, for example, Ruscic [5] and Ruscic and Bross[6]).
Note that an uncertainty of ± 0.000 kJ/mol indicates that the estimated uncertainty is < ± 0.0005 kJ/mol.
|
|
|
Website Functionality Credits
|
The reorganization of the website was developed and implemented by David H. Bross (ANL).
The find function is based on the complete Species Dictionary entries for the appropriate version of the ATcT TN.
The molecule images are rendered by Indigo-depict.
The XYZ renderings are based on Jmol: an open-source Java viewer for chemical structures in 3D. http://www.jmol.org/.
|
|
|
Acknowledgement
|
|
This work was supported by the U.S. Department of Energy, Office of Science, Office of Basic Energy Sciences, Division of Chemical Sciences, Geosciences and Biosciences under Contract No. DE-AC02-06CH11357.
|





![c1c(cc2ccccc2c1)[H+]](../images/2992.png)

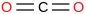
=O](../images/488.png)

