Selected ATcT [1, 2] enthalpy of formation based on version 1.124 of the Thermochemical Network [3] This version of ATcT results was generated by additional expansion of version 1.122x [4] to include additional information relevant to the study of thermophysical and thermochemical properties of CH2 and CH3 using nonrigid rotor anharmonic oscillator (NRRAO) partition functions [5], the development and benchmarking of a state-of-the-art computational approach that aims to reproduce total atomization energies of small molecules within 10–15 cm-1 [6], as well as the study of the reversible reaction C2H3 + H2 ⇌ C2H4 + H ⇌ C2H5 [7]
|
Formic acid | | Formula: HC(O)OH (g, anti) | | CAS RN: 64-18-6 | | ATcT ID: 64-18-6*2 | | SMILES: C(=O)O | | InChI: InChI=1S/CH2O2/c2-1-3/h1H,(H,2,3) | | InChIKey: BDAGIHXWWSANSR-UHFFFAOYSA-N | | Hills Formula: C1H2O2 | 2D Image: | 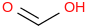 | | Aliases: HC(O)OH; Formic acid; Hydrogen carboxylic acid; Methanoic acid; HCOOH; OCHOH; O=CH-OH; H(C=O)OH; Formylic acid; Aminic acid | | Relative Molecular Mass: 46.0254 ± 0.0010 |
| ΔfH°(0 K) | ΔfH°(298.15 K) | Uncertainty | Units |
|---|
| -354.72 | -361.82 | ± 0.38 | kJ/mol |
| 3D Image of HC(O)OH (g, anti) |
 loadScript ./j2s/core/package.js loadScript ./j2s/core/corejmol.z.js JSmol exec jmolApplet0 start applet null Jmol JavaScript applet jmolApplet0__4186146611533__ initializing Jmol getValue debug null Jmol getValue logLevel null Jmol getValue allowjavascript true AppletRegistry.checkIn(jmolApplet0__4186146611533__) vwrOptions: { "name":"jmolApplet0","allowJavaScript":true,"applet":true,"documentBase":"https://atct.anl.gov/Thermochemical%20Data/version%201.124/species/?species_number=218","platform":"J.awtjs2d.Platform","fullName":"jmolApplet0__4186146611533__","codePath":"https://atct.anl.gov/Thermochemical%20Data/version%201.124/species/./j2s/","display":"jmolApplet0_canvas2d","signedApplet":"true","appletReadyCallback":"Jmol._readyCallback","statusListener":"[J.appletjs.Jmol object]","syncId":"4186146611533","bgcolor":"#FFFFFF" } setting document base to "https://atct.anl.gov/Thermochemical%20Data/version%201.124/species/?species_number=218" (C) 2015 Jmol Development Jmol Version: 14.6.1_2016.08.11 2016-08-11 22:11 java.vendor: Java2Script (HTML5) java.version: 2016-08-02 04:55:11 (JSmol/j2s) os.name: Mozilla/5.0 AppleWebKit/537.36 (KHTML, like Gecko; compatible; ClaudeBot/1.0; +claudebot@anthropic.com) Access: ALL memory: 0.0/0.0 processors available: 1 useCommandThread: false appletId:jmolApplet0 (signed) loadScript ./j2s/core/corescript.z.js Jmol getValue emulate null defaults = "Jmol" Jmol getValue boxbgcolor null Jmol getValue bgcolor #FFFFFF backgroundColor = "#FFFFFF" Jmol getValue ANIMFRAMECallback null Jmol getValue APPLETREADYCallback Jmol._readyCallback APPLETREADYCallback = "Jmol._readyCallback" callback set for APPLETREADYCallback Jmol._readyCallback APPLETREADY Jmol getValue ATOMMOVEDCallback null Jmol getValue CLICKCallback null Jmol getValue DRAGDROPCallback null Jmol getValue ECHOCallback null Jmol getValue ERRORCallback null Jmol getValue EVALCallback null Jmol getValue HOVERCallback null Jmol getValue IMAGECallback null Jmol getValue LOADSTRUCTCallback null Jmol getValue MEASURECallback null Jmol getValue MESSAGECallback null Jmol getValue MINIMIZATIONCallback null Jmol getValue SERVICECallback null Jmol getValue PICKCallback null Jmol getValue RESIZECallback null Jmol getValue SCRIPTCallback null Jmol getValue SYNCCallback null Jmol getValue STRUCTUREMODIFIEDCallback null Jmol getValue doTranslate null language=en_US Jmol getValue popupMenu null Jmol getValue script null Jmol getValue loadInline null Jmol getValue load null Jmol applet jmolApplet0__4186146611533__ ready script 1 started zoomLarge = false antialiasDisplay = true FileManager.getAtomSetCollectionFromFile(../xyz/HC(O)OH_(anti).xyz) FileManager opening url https://atct.anl.gov/Thermochemical%20Data/version%201.124/xyz/HC(O)OH_(anti).xyz The Resolver thinks Xyz anti-Formic acid 1A' Cs B2PLYPD3/aug-cc-pVTZ anti-Formic acid 1A' Cs B3LYP/cc-pVTZ+d anti-Formic acid 1A' Cs B3LYP/6-31G(2df,p) Time for openFile(../xyz/HC(O)OH_(anti).xyz): 128 ms reading 15 atoms ModelSet: haveSymmetry:false haveUnitcells:false haveFractionalCoord:false 3 models in this collection. Use getProperty "modelInfo" or getProperty "auxiliaryInfo" to inspect them. Default Van der Waals type for model set to Babel 15 atoms created ModelSet: autobonding; use autobond=false to not generate bonds automatically Time for creating model: 11 ms anti-Formic acid 1A' Cs B2PLYPD3/aug-cc-pVTZ anti-Formic acid 1A' Cs B3LYP/cc-pVTZ+d anti-Formic acid 1A' Cs B3LYP/6-31G(2df,p) 3 models loadScript ./j2s/J/thread/SpinThread.js Script completed Jmol script terminated | | spin ON spin OFF | | | Top contributors to the provenance of ΔfH° of HC(O)OH (g, anti)The 20 contributors listed below account only for 77.3% of the provenance of ΔfH° of HC(O)OH (g, anti).
A total of 78 contributors would be needed to account for 90% of the provenance.
Please note: The list is limited to 20 most important contributors or, if less, a number sufficient to account for 90% of the provenance. The Reference acts as a further link to the relevant references and notes for the measurement. The Measured Quantity is normaly given in the original units; in cases where we have reinterpreted the original measurement, the listed value may differ from that given by the authors. The quoted uncertainty is the a priori uncertainty used as input when constructing the initial Thermochemical Network, and corresponds either to the value proposed by the original authors or to our estimate; if an additional multiplier is given in parentheses immediately after the prior uncertainty, it corresponds to the factor by which the prior uncertainty needed to be multiplied during the ATcT analysis in order to make that particular measurement consistent with the prevailing knowledge contained in the Thermochemical Network.
| Contribution
(%) | TN
ID | Reaction | Measured Quantity | Reference | | 52.6 | 4309.1 | HC(O)OH (g, syn) → HC(O)OH (g, anti) | ΔrH°(0 K) = 1365 ± 30 cm-1 | Hocking 1976, Gurvich TPIS | | 4.9 | 4316.2 | HC(O)OH (cr,l) + 1/2 O2 (g) → CO2 (g) + H2O (cr,l) | ΔrH°(298.15 K) = -60.807 ± 0.074 kcal/mol | Lebedeva 1964 | | 3.4 | 4316.1 | HC(O)OH (cr,l) + 1/2 O2 (g) → CO2 (g) + H2O (cr,l) | ΔrH°(298.15 K) = -60.851 ± 0.089 kcal/mol | Sinke 1959 | | 3.2 | 4317.1 | HC(O)OH (cr,l) → HC(O)OH (g) | ΔrH°(298.15 K) = 46.30 ± 0.23 kJ/mol | Majer 1985 | | 1.1 | 2974.2 | HC(O)OH (g, syn) → [HCO]+ (g) + OH (g) | ΔrH°(0 K) = 12.81 ± 0.01 eV | Traeger 1985, AE corr | | 1.0 | 4317.2 | HC(O)OH (cr,l) → HC(O)OH (g) | ΔrH°(298.15 K) = 46.15 ± 0.40 kJ/mol | NBS Tables 1989 | | 1.0 | 4315.2 | HC(O)OH (g) → [HCO]+ (g) + OH (g) | ΔrH°(0 K) = 1235.7 ± 1.0 kJ/mol | Shuman 2010a, Bomble 2006 | | 0.9 | 4314.8 | HC(O)OH (g, anti) + CH4 (g) → CH2O (g) + CH3OH (g) | ΔrH°(0 K) = 29.70 ± 0.9 kcal/mol | Ruscic W1RO | | 0.8 | 4602.8 | CH3C(O)OH (g, syn) + HC(O)OH (g, anti) → CH3C(O)OH (g, anti) + HC(O)OH (g, syn) | ΔrH°(0 K) = 346 ± 200 cm-1 | Ruscic W1RO | | 0.7 | 4296.1 | HC(O)OH (g, anti) → [HC(O)OH]+ (g, syn) | ΔrH°(0 K) = 11.16 ± 0.03 eV | Warneck 1974 | | 0.7 | 4296.2 | HC(O)OH (g, anti) → [HC(O)OH]+ (g, syn) | ΔrH°(0 K) = 11.16 ± 0.03 eV | Matthews 1969 | | 0.7 | 4602.4 | CH3C(O)OH (g, syn) + HC(O)OH (g, anti) → CH3C(O)OH (g, anti) + HC(O)OH (g, syn) | ΔrH°(0 K) = 395 ± 210 cm-1 | Ruscic G4 | | 0.7 | 4602.7 | CH3C(O)OH (g, syn) + HC(O)OH (g, anti) → CH3C(O)OH (g, anti) + HC(O)OH (g, syn) | ΔrH°(0 K) = 355 ± 210 cm-1 | Ruscic CBS-n | | 0.7 | 4291.3 | HC(O)OH (g, syn) → C (g) + 2 H (g) + 2 O (g) | ΔrH°(0 K) = 479.81 ± 0.30 kcal/mol | Karton 2011 | | 0.7 | 4314.7 | HC(O)OH (g, anti) + CH4 (g) → CH2O (g) + CH3OH (g) | ΔrH°(0 K) = 30.25 ± 1.0 kcal/mol | Ruscic CBS-n | | 0.7 | 4314.4 | HC(O)OH (g, anti) + CH4 (g) → CH2O (g) + CH3OH (g) | ΔrH°(0 K) = 29.55 ± 1.0 kcal/mol | Ruscic G4 | | 0.6 | 4317.3 | HC(O)OH (cr,l) → HC(O)OH (g) | ΔrH°(298.15 K) = 46.3 ± 0.5 kJ/mol | Konicek 1970 | | 0.6 | 4602.3 | CH3C(O)OH (g, syn) + HC(O)OH (g, anti) → CH3C(O)OH (g, anti) + HC(O)OH (g, syn) | ΔrH°(0 K) = 412 ± 225 cm-1 | Ruscic G3X | | 0.6 | 4314.3 | HC(O)OH (g, anti) + CH4 (g) → CH2O (g) + CH3OH (g) | ΔrH°(0 K) = 29.74 ± 1.1 kcal/mol | Ruscic G3X | | 0.6 | 4601.9 | CH3C(O)OH (g, syn) → CH3C(O)OH (g, anti) | ΔrH°(0 K) = 20.72 ± 0.50 kJ/mol | Feller 2018a, est unc |
| Top 10 species with enthalpies of formation correlated to the ΔfH° of HC(O)OH (g, anti) | Please note: The correlation coefficients are obtained by renormalizing the off-diagonal elements of the covariance matrix by the corresponding variances.
The correlation coefficient is a number from -1 to 1, with 1 representing perfectly correlated species, -1 representing perfectly anti-correlated species, and 0 representing perfectly uncorrelated species.
| Correlation
Coefficent
(%) | Species Name | Formula | Image | ΔfH°(0 K) | ΔfH°(298.15 K) | Uncertainty | Units | Relative
Molecular
Mass | ATcT ID | | 53.1 | Formic acid | HC(O)OH (g, syn) | 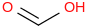 | -371.05 | -378.37 | ± 0.22 | kJ/mol | 46.0254 ±
0.0010 | 64-18-6*1 | | 53.1 | Formic acid | HC(O)OH (g) | 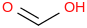 | -371.05 | -378.34 | ± 0.22 | kJ/mol | 46.0254 ±
0.0010 | 64-18-6*0 | | 45.6 | Formic acid cation | [HC(O)OH]+ (g, syn) | O](../images/691.png) | 721.93 | 714.84 | ± 0.25 | kJ/mol | 46.0248 ±
0.0010 | 50614-05-6*1 | | 45.6 | Formic acid cation | [HC(O)OH]+ (g) | O](../images/2637.png) | 721.93 | 715.40 | ± 0.25 | kJ/mol | 46.0248 ±
0.0010 | 50614-05-6*0 | | 35.6 | Formic acid | HC(O)OH (cr,l) | 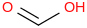 | -431.51 | -424.67 | ± 0.20 | kJ/mol | 46.0254 ±
0.0010 | 64-18-6*500 | | 15.8 | Formate | [HC(O)O]- (aq) | ![C(=O)[O-]](../images/2683.png) | | -425.48 | ± 0.45 | kJ/mol | 45.0180 ±
0.0010 | 71-47-6*800 | | 15.8 | Formic acid | HC(O)OH (aq) | 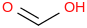 | | -425.48 | ± 0.45 | kJ/mol | 46.0254 ±
0.0010 | 64-18-6*800 | | 15.8 | Formic acid | HC(O)OH (aq, undissoc) | 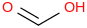 | | -425.38 | ± 0.45 | kJ/mol | 46.0254 ±
0.0010 | 64-18-6*1000 | | 14.2 | Carbonic acid | C(O)(OH)2 (g, cis-cis) | 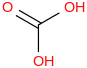 | -602.53 | -612.70 | ± 0.75 | kJ/mol | 62.0248 ±
0.0012 | 463-79-6*1 | | 14.2 | Carbonic acid | C(O)(OH)2 (g) | 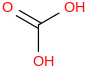 | -602.53 | -611.84 | ± 0.75 | kJ/mol | 62.0248 ±
0.0012 | 463-79-6*0 |
| Most Influential reactions involving HC(O)OH (g, anti)Please note: The list, which is based on a hat (projection) matrix analysis, is limited to no more than 20 largest influences.
| Influence
Coefficient | TN
ID | Reaction | Measured Quantity | Reference | | 0.774 | 4309.1 | HC(O)OH (g, syn) → HC(O)OH (g, anti) | ΔrH°(0 K) = 1365 ± 30 cm-1 | Hocking 1976, Gurvich TPIS | | 0.145 | 4299.7 | [HC(O)OH]- (g, anti) → HC(O)OH (g, anti) | ΔrH°(0 K) = -1.183 ± 0.050 eV | Ruscic W1RO | | 0.117 | 4699.5 | CH3C(O)OCH3 (g, anti) + HC(O)OH (g, anti) → CH3C(O)OH (g, anti) + HC(O)OCH3 (g, anti) | ΔrH°(0 K) = -1.61 ± 0.85 kcal/mol | Ruscic W1RO | | 0.105 | 4699.1 | CH3C(O)OCH3 (g, anti) + HC(O)OH (g, anti) → CH3C(O)OH (g, anti) + HC(O)OCH3 (g, anti) | ΔrH°(0 K) = -1.37 ± 0.90 kcal/mol | Ruscic G3X | | 0.105 | 4699.2 | CH3C(O)OCH3 (g, anti) + HC(O)OH (g, anti) → CH3C(O)OH (g, anti) + HC(O)OCH3 (g, anti) | ΔrH°(0 K) = -1.38 ± 0.90 kcal/mol | Ruscic G4 | | 0.105 | 4699.4 | CH3C(O)OCH3 (g, anti) + HC(O)OH (g, anti) → CH3C(O)OH (g, anti) + HC(O)OCH3 (g, anti) | ΔrH°(0 K) = -1.54 ± 0.90 kcal/mol | Ruscic CBS-n | | 0.097 | 4299.4 | [HC(O)OH]- (g, anti) → HC(O)OH (g, anti) | ΔrH°(0 K) = -1.161 ± 0.061 eV | Ruscic G4 | | 0.097 | 4297.8 | HC(O)OH (g, anti) → [HC(O)OH]+ (g, anti) | ΔrH°(0 K) = 11.188 ± 0.040 eV | Ruscic W1RO | | 0.085 | 4699.3 | CH3C(O)OCH3 (g, anti) + HC(O)OH (g, anti) → CH3C(O)OH (g, anti) + HC(O)OCH3 (g, anti) | ΔrH°(0 K) = -1.47 ± 1.00 kcal/mol | Ruscic CBS-n | | 0.050 | 4299.3 | [HC(O)OH]- (g, anti) → HC(O)OH (g, anti) | ΔrH°(0 K) = -1.217 ± 0.085 eV | Ruscic G3X | | 0.045 | 4732.5 | HC(O)OH (g, anti) + CH3CH2OH (g) → HC(O)OCH2CH3 (g, anti) + H2O (g) | ΔrH°(0 K) = -4.88 ± 1.50 kcal/mol | Ruscic W1RO | | 0.043 | 4299.6 | [HC(O)OH]- (g, anti) → HC(O)OH (g, anti) | ΔrH°(0 K) = -1.190 ± 0.092 eV | Ruscic CBS-n | | 0.041 | 4602.8 | CH3C(O)OH (g, syn) + HC(O)OH (g, anti) → CH3C(O)OH (g, anti) + HC(O)OH (g, syn) | ΔrH°(0 K) = 346 ± 200 cm-1 | Ruscic W1RO | | 0.040 | 4732.4 | HC(O)OH (g, anti) + CH3CH2OH (g) → HC(O)OCH2CH3 (g, anti) + H2O (g) | ΔrH°(0 K) = -5.37 ± 1.60 kcal/mol | Ruscic CBS-n | | 0.040 | 4732.2 | HC(O)OH (g, anti) + CH3CH2OH (g) → HC(O)OCH2CH3 (g, anti) + H2O (g) | ΔrH°(0 K) = -4.91 ± 1.60 kcal/mol | Ruscic G4 | | 0.037 | 4602.7 | CH3C(O)OH (g, syn) + HC(O)OH (g, anti) → CH3C(O)OH (g, anti) + HC(O)OH (g, syn) | ΔrH°(0 K) = 355 ± 210 cm-1 | Ruscic CBS-n | | 0.037 | 4602.4 | CH3C(O)OH (g, syn) + HC(O)OH (g, anti) → CH3C(O)OH (g, anti) + HC(O)OH (g, syn) | ΔrH°(0 K) = 395 ± 210 cm-1 | Ruscic G4 | | 0.034 | 4732.1 | HC(O)OH (g, anti) + CH3CH2OH (g) → HC(O)OCH2CH3 (g, anti) + H2O (g) | ΔrH°(0 K) = -5.23 ± 1.72 kcal/mol | Ruscic G3X | | 0.032 | 4602.3 | CH3C(O)OH (g, syn) + HC(O)OH (g, anti) → CH3C(O)OH (g, anti) + HC(O)OH (g, syn) | ΔrH°(0 K) = 412 ± 225 cm-1 | Ruscic G3X | | 0.029 | 4297.4 | HC(O)OH (g, anti) → [HC(O)OH]+ (g, anti) | ΔrH°(0 K) = 11.143 ± 0.073 eV | Ruscic G4 |
|
|
|
References
|
|
1
|
|
B. Ruscic, R. E. Pinzon, M. L. Morton, G. von Laszewski, S. Bittner, S. G. Nijsure, K. A. Amin, M. Minkoff, and A. F. Wagner,
Introduction to Active Thermochemical Tables: Several "Key" Enthalpies of Formation Revisited.
J. Phys. Chem. A 108, 9979-9997 (2004)
[DOI: 10.1021/jp047912y]
|
|
2
|
|
B. Ruscic, R. E. Pinzon, G. von Laszewski, D. Kodeboyina, A. Burcat, D. Leahy, D. Montoya, and A. F. Wagner,
Active Thermochemical Tables: Thermochemistry for the 21st Century.
J. Phys. Conf. Ser. 16, 561-570 (2005)
[DOI: 10.1088/1742-6596/16/1/078]
|
|
3
|
|
B. Ruscic and D. H. Bross,
Active Thermochemical Tables (ATcT) values based on ver. 1.124 of the Thermochemical Network, Argonne National Laboratory, Lemont, Illinois 2022; available at ATcT.anl.gov
[DOI: 10.17038/CSE/1885923]
|
|
4
|
|
Y. Ren, L. Zhou, A. Mellouki, V. Daële, M. Idir, S. S. Brown, B. Ruscic, Robert S. Paton, M. R. McGillen, and A. R. Ravishankara,
Reactions of NO3 with Aromatic Aldehydes: Gas-Phase Kinetics and Insights into the Mechanism of the Reaction.
Atmos. Chem. Phys. 21, 13537-13551 (2021)
[DOI: 10.5194/acp2021-228]
|
|
5
|
|
B. Ruscic and D. H. Bross,
Active Thermochemical Tables: The Thermophysical and Thermochemical Properties of Methyl, CH3, and Methylene, CH2, Corrected for Nonrigid Rotor and Anharmonic Oscillator Effects.
Mol. Phys. e1969046 (2021)
[DOI: 10.1080/00268976.2021.1969046]
|
|
6
|
|
J. H. Thorpe, J. L. Kilburn, D. Feller, P. B. Changala, D. H. Bross, B. Ruscic, and J. F. Stanton,
Elaborated Thermochemical Treatment of HF, CO, N2, and H2O: Insight into HEAT and Its Extensions
J. Chem. Phys. 155, 184109 (2021)
[DOI: 10.1063/5.0069322]
|
|
7
|
|
T. L. Nguyen, D. H. Bross, B. Ruscic, G. B. Ellison, and J. F. Stanton,
Mechanism, Thermochemistry, and Kinetics of the Reversible Reactions: C2H3 + H2 ⇌ C2H4 + H ⇌ C2H5.
Faraday Discuss. , (Advance Article) (2022)
[DOI: 10.1039/D1FD00124H]
|
|
8
|
|
B. Ruscic,
Uncertainty Quantification in Thermochemistry, Benchmarking Electronic Structure Computations, and Active Thermochemical Tables.
Int. J. Quantum Chem. 114, 1097-1101 (2014)
[DOI: 10.1002/qua.24605]
|
|
9
|
|
B. Ruscic and D. H. Bross,
Thermochemistry
Computer Aided Chem. Eng. 45, 3-114 (2019)
[DOI: 10.1016/B978-0-444-64087-1.00001-2]
|
|
|
|
Formula
|
|
The aggregate state is given in parentheses following the formula, such as: g - gas-phase, cr - crystal, l - liquid, etc.
|
|
|
Uncertainties
|
The listed uncertainties correspond to estimated 95% confidence limits, as customary in thermochemistry (see, for example, Ruscic [8,9]).
Note that an uncertainty of ± 0.000 kJ/mol indicates that the estimated uncertainty is < ± 0.0005 kJ/mol.
|
|
|
Website Functionality Credits
|
The reorganization of the website was developed and implemented by David H. Bross (ANL).
The find function is based on the complete Species Dictionary entries for the appropriate version of the ATcT TN.
The molecule images are rendered by Indigo-depict.
The XYZ renderings are based on Jmol: an open-source Java viewer for chemical structures in 3D. http://www.jmol.org/.
|
|
|
Acknowledgement
|
|
This work was supported by the U.S. Department of Energy, Office of Science, Office of Basic Energy Sciences, Division of Chemical Sciences, Geosciences and Biosciences under Contract No. DE-AC02-06CH11357.
|
|
Page generated: 03/29/2025
|







