Selected ATcT [1, 2] enthalpy of formation based on version 1.122 of the Thermochemical Network [3]
This version of ATcT results was partially described in Ruscic et al. [4], and was also used for the initial development of high-accuracy ANLn composite electronic structure methods [5].| Species Name | Formula | Image | ΔfH°(0 K) | ΔfH°(298.15 K) | Uncertainty | Units | Relative Molecular Mass |
ATcT ID |
|---|---|---|---|---|---|---|---|---|
| Ethylene | C2H4 (g) |  | 60.96 | 52.45 | ± 0.13 | kJ/mol | 28.0532 ± 0.0016 | 74-85-1*0 |
| Formaldehyde | H2CO (g) | 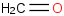 | -105.349 | -109.188 | ± 0.099 | kJ/mol | 30.02598 ± 0.00087 | 50-00-0*0 |
| Difluorophosgene | COF2 (g) |  | -603.50 | -606.59 | ± 0.46 | kJ/mol | 66.00691 ± 0.00085 | 353-50-4*0 |
| Tetrafluoroethylene | C2F4 (g) |  | -671.52 | -674.93 | ± 0.57 | kJ/mol | 100.0150 ± 0.0016 | 116-14-3*0 |
Reaction Enthalpy
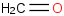 +
+  →
→  + 2
+ 2 
Note: The listed uncertainity of the reaction enthalpy is computed using the full covariance matrix.
| 1 |
B. Ruscic, R. E. Pinzon, M. L. Morton, G. von Laszewski, S. Bittner, S. G. Nijsure, K. A. Amin, M. Minkoff, and A. F. Wagner, Introduction to Active Thermochemical Tables: Several "Key" Enthalpies of Formation Revisited. J. Phys. Chem. A 108, 9979-9997 (2004) [DOI: 10.1021/jp047912y] |
|
| 2 |
B. Ruscic, R. E. Pinzon, G. von Laszewski, D. Kodeboyina, A. Burcat, D. Leahy, D. Montoya, and A. F. Wagner, Active Thermochemical Tables: Thermochemistry for the 21st Century. J. Phys. Conf. Ser. 16, 561-570 (2005) [DOI: 10.1088/1742-6596/16/1/078] |
|
| 3 |
B. Ruscic and D. H. Bross, Active Thermochemical Tables (ATcT) values based on ver. 1.122 of the Thermochemical Network (2016); available at ATcT.anl.gov |
|
| 4 |
B. Ruscic, Active Thermochemical Tables: Sequential Bond Dissociation Enthalpies of Methane, Ethane, and Methanol and the Related Thermochemistry. J. Phys. Chem. A 119, 7810-7837 (2015) [DOI: 10.1021/acs.jpca.5b01346] |
|
| 5 |
S. J. Klippenstein, L. B. Harding, and B. Ruscic, Ab initio Computations and Active Thermochemical Tables Hand in Hand: Heats of Formation of Core Combustion Species. J. Phys. Chem. A 121, 6580-6602 (2017) [DOI: 10.1021/acs.jpca.7b05945] |
|
| 6 |
B. Ruscic, Uncertainty Quantification in Thermochemistry, Benchmarking Electronic Structure Computations, and Active Thermochemical Tables. Int. J. Quantum Chem. 114, 1097-1101 (2014) [DOI: 10.1002/qua.24605] |
Note that an uncertainty of ± 0.000 kJ/mol indicates that the estimated uncertainty is < ± 0.0005 kJ/mol.
The find function is based on the complete Species Dictionary entries for the appropriate version of the ATcT TN.
The molecule images are rendered by Indigo-depict.
The XYZ renderings are based on Jmol: an open-source Java viewer for chemical structures in 3D. http://www.jmol.org/.